

Description
This unique product - which we developed in close collaboration with its inventors at Stanford University - offers a range of truly exciting surface immobilization schemes. SAMs terminated with azide groups react with acetylene-bearing molecules through the so-called Sharpless "click" chemistry (Angew. Chem. Int. Ed. 2002, 41, 2596-2599) to form stable triazole linkages. In a series of ingenious experiments, the Stanford team showed (i) how this reaction can be used to immobilize various (bio)molecules chemoselectively and in excellent yields (J. Am. Chem. Soc. 2005, 127, 8600-8601), and (ii) how it can be coupled with electrochemistry to allow for site-selective immobilization (J. Am. Chem. Soc. 2006, 128, 1794-1795 and also Science, Editor?s Choice, 10 February, 2006).
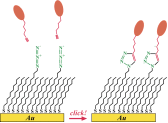
In the latter case, application of voltage bias to a gold microelectrode supporting azide-terminated SAM changed the redox state of an auxiliary copper catalyst, and thus activated the SAM for the "click" chemistry. By activating different microelectrodes at different times, it was possible to immobilize various molecules onto them. The capability of such sequential immobilization should prove extremely useful in the development of electrochemical microsensor arrays.

Note: Please see our related products for inclusion in bioresistant monolayers on gold (HS-Cn-EGm-N3; cat # TH 008 under Thiols for Bioresistant and Biospecific SAMs) and silanes for functionalization of glass, Si and PDMS (cat # SI 004 - SI 006 under Silanes).
Standard Products
| name | catalog no. | m | n | weight | price |
|---|---|---|---|---|---|
| HS-C11N3 | FT 010-m11-0.2 | 11 | 200 mg | Log in to see the prices | |
| HS-C11N3 | FT 010-m11-0.5 | 11 | 500 mg | ||
| HS-C11N3 | FT 010-m11-1 | 11 | 1 g |
Custom synthesis variants
This product is also available on custom synthesis basis, in the following variants:
m: 10, 12, 16, 17
n: n/a
Note, if none of the listed structures fit your specific requirements, please submit your own structure formula within our custom synthesis contact form by clicking button below.